Researchers at Stanford University and the US Department of Energy’s SLAC National Accelerator Laboratory have explored the potential recovery of lost capacity in lithium batteries by using an extremely fast discharging step to reconnect an island of inactive lithium with the anode. Adding this extra step slowed the degradation of their test battery and increased its lifespan by nearly 30%.
One of the main reasons for substantial capacity losses and even fires in lithium batteries are little islands of inactive lithium that are created during the nonuniform dissolution of lithium dendrites. This isolated lithium loses connection with the current collector, so it is considered electrochemically inactive or “dead,” but a team of US researchers has discovered that this lithium can be brought back to life, boosting the capacity and lifespan of batteries.
Researchers at the US Department of Energy’s SLAC National Accelerator Laboratory and Stanford University have shown that isolated lithium is alive and highly responsive to battery operations, due to its dynamic polarization to the electric field in the electrolyte. They have designed an optical cell with a lithium-nickel-manganese-cobalt-oxide (NMC) cathode and a lithium anode. They also used an isolated lithium island in between as a test device that allowed them to track what happens inside a battery when in use, in real time.
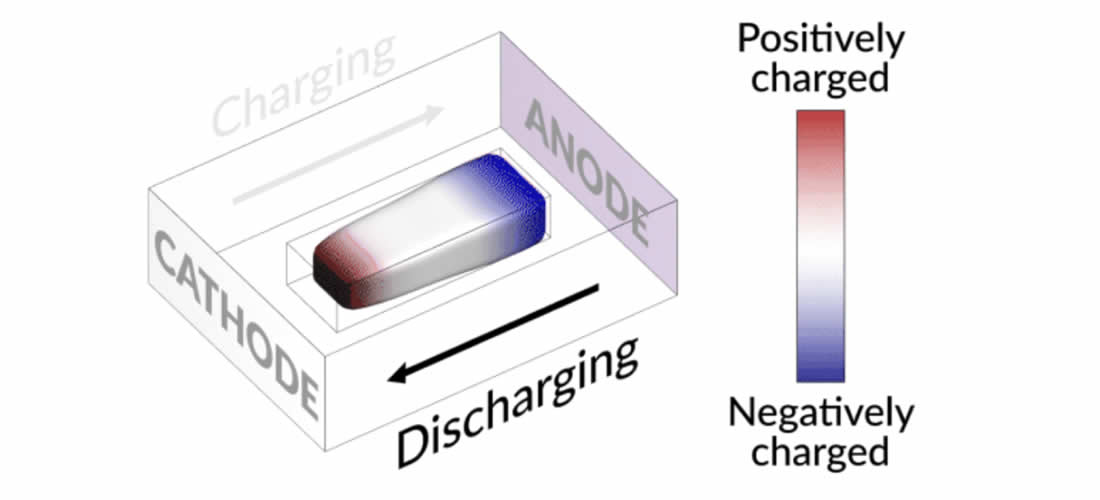
They discovered that when charging the cell, the island slowly moved toward the cathode. When discharging, it crept in the opposite direction.
“It’s like a very slow worm that inches its head forward and pulls its tail in to move nanometer by nanometer,” said Yi Cui, a professor at Stanford University and SLAC, and an investigator at the Stanford Institute for Materials and Energy Research. “In this case, it transports by dissolving away on one end and depositing material to the other end. If we can keep the lithium worm moving, it will eventually touch the anode and reestablish the electrical connection.”
Lithium metal accumulates at the negative end of the island and dissolves at the positive end. This continual growth and dissolution causes the back-and-forth movement that the researchers observed. Their next step was to modify the charging protocol in order to move these floating islands far enough to reconnect with the anode.
They discovered that by adding a brief, high-current discharging step right after charging, the battery nudges the island to grow in the direction of the anode. As a result, the researchers demonstrated that they can mobilize and recover the isolated lithium, extending the battery’s lifespan by nearly 30%.
The researchers expect these findings to have broad implications for the design and development of more robust lithium-metal batteries. Their study, “Dynamic spatial progression of isolated lithium during battery operations,” was recently published in Nature.
Source: www.pv-magazine.com
 En
En